Key Points:
- Scientists captured detailed images of a DNA repair process that could help fight cancer.
- The discovery focuses on stopping the RAD52 protein, which helps cancer cells with BRCA mutations survive.
- Researchers used an ancient yeast protein called Mgm101 to study how this repair mechanism actually works.
- The team used advanced imaging tools to observe the protein ring binding and fixing broken DNA strands.
Scientists just achieved a breakthrough in cancer research by capturing the most detailed structural images of a specific DNA repair process. This impressive discovery could eventually reveal new ways to stop the dangerous effects of BRCA1 and BRCA2 gene mutations. Doctors know these specific mutations greatly increase the risk of breast, ovarian, and several other deadly cancers. By understanding exactly how cells fix their broken DNA, medical researchers can design better drugs to destroy cancer cells before they multiply.
Cancer cells often survive because they find clever ways to fix their own damage. Previous research showed that a human protein called RAD52 performs critical DNA repair in cancer cells that lack normal BRCA genes. Healthy BRCA genes naturally suppress tumors, but when they mutate, cells rely heavily on RAD52 to stay alive and replicate. Scientists realized that if they could successfully block the RAD52 protein, they could effectively kill these vulnerable cancer cells.
However, blocking this specific protein requires a complete understanding of how it actually works. Until now, researchers have struggled to capture RAD52 repair activities because human proteins are highly complex. Even the most sophisticated imaging techniques failed to show the full process. To solve this problem, the research team decided to look back in time. They turned their attention to an ancient ancestral protein called Mgm101, which lives in yeast mitochondria.
By studying this simpler yeast protein, the team observed several key steps in a repair process called single-strand DNA annealing. Charles Bell, a professor of biological chemistry and pharmacology at The Ohio State University College of Medicine, led the groundbreaking study. He explained that getting a clearer picture of how this protein family binds to broken DNA strands and coaxes them back together provides vital clues for future drug targets.
Bell noted that while researchers still consider this a proposed mechanism, they now possess the best snapshots ever taken of any protein performing single-strand annealing. He emphasized that these detailed images will strongly guide future strategies for drug development. The team recently published their exciting findings in the well-known journal Nucleic Acids Research, sharing their structural images with the global scientific community.
DNA strands break constantly in every single cell of the human body. Proteins must work quickly to fix these breaks and keep cellular processes running smoothly. Because this repair happens so incredibly fast, taking clear pictures of the event presents a massive challenge. Bell partnered with Vicki Wysocki, a professor emerita at Ohio State and chair of the School of Chemistry and Biochemistry at the Georgia Institute of Technology, to tackle this imaging problem.
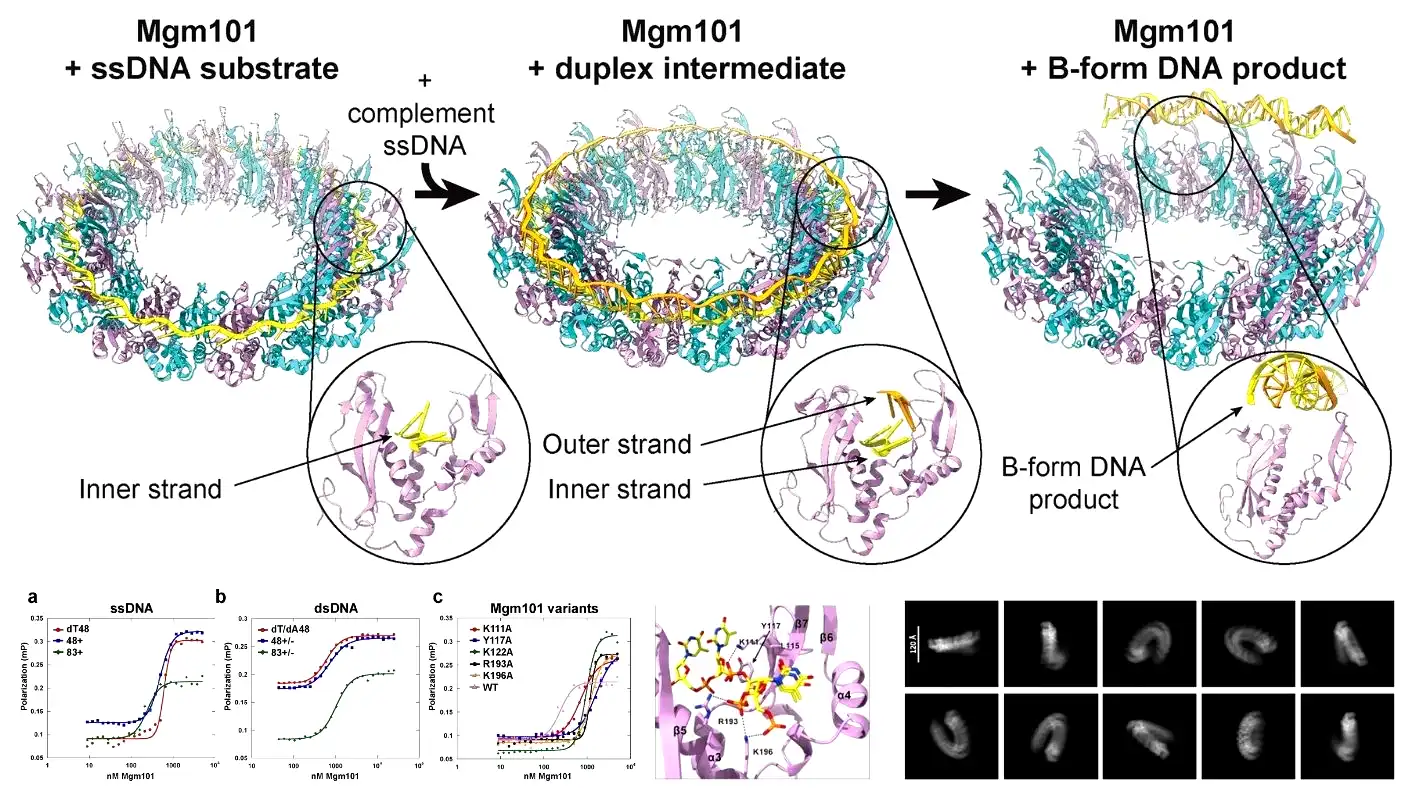
Wysocki brought specialized expertise to the project. Her laboratory uses native mass spectrometry and mass photometry, which rely on light to measure the exact masses of protein and DNA complexes. Using these advanced tools, the team discovered how the Mgm101 protein actually builds itself. They found that the protein assembled from a single copy into a massive multi-unit molecular complex. They call this complex a 19-mer because it forms a distinct ring made up of exactly 19 copies of the protein.
Wysocki explained how this ring functions during the repair process. The protein ring sits patiently as a physical template. The 1st broken DNA strand comes down and attaches to the ring. Soon after, the 2nd strand arrives and begins annealing, joining seamlessly with the 1st strand. Bell and his team supported these findings by using cryogenic electron microscopy. They froze structures in an ultra-thin layer of ice and observed them floating in solution.
These high-resolution structures finally revealed multiple phases of the repair process that nobody had ever seen before. The images showed the initial substrate phase, in which the 19-mer ring contains only a single DNA strand. Next, they captured the duplex intermediate phase, showing the 2nd strand moving into place for annealing. Finally, they photographed the B-form product, which shows fully repaired DNA releasing in a classic double-helix formation.
Bell pointed out that previous researchers only managed to capture RAD52 structures holding single-stranded DNA. Nobody had ever caught the protein holding the 2 distinct DNA strands it was actively trying to anneal. The new study successfully mapped out the full pathway from start to finish. More importantly, the team captured the duplex intermediate conformation for the very first time.
During this newly discovered intermediate phase, the protein binds the 1st DNA strand completely around the ring. It grabs the DNA only by its sugar-phosphate backbone. This specific grip leaves the nucleotide bases pointing straight up, fully exposed, and completely separated. The DNA stretches out and unwinds into a perfect circle so the protein can easily search for the matching genetic sequence.
Before this study, scientists debated whether 1 or 2 protein rings were responsible for this complex repair mechanism. The new findings clearly prove that a single molecular complex handles the entire job. Moving forward, the research team plans to capture these same phases using the human RAD52 protein. They want to focus heavily on the duplex intermediate phase and use mass spectrometry to learn even more about how DNA binds to these life-saving proteins.
Source: Nucleic Acids Research (2026).