Key Points:
- A research team created a new imaging method to watch genes act inside an embryo.
- The technology allows scientists to see the activity of thousands of genes at once.
- Researchers built a comprehensive 4D atlas detailing early embryonic development.
- The data shows how cells change their genetic programming to form clear tissue boundaries.
A tiny cluster of cells somehow knows exactly how to build a complex living creature with a head, trunk, and tail. For decades, scientists have struggled to fully understand how thousands of genes coordinate this incredible feat of biological engineering. Now, thanks to a groundbreaking new imaging method, researchers can finally watch this process happen in real time across an entire embryo.
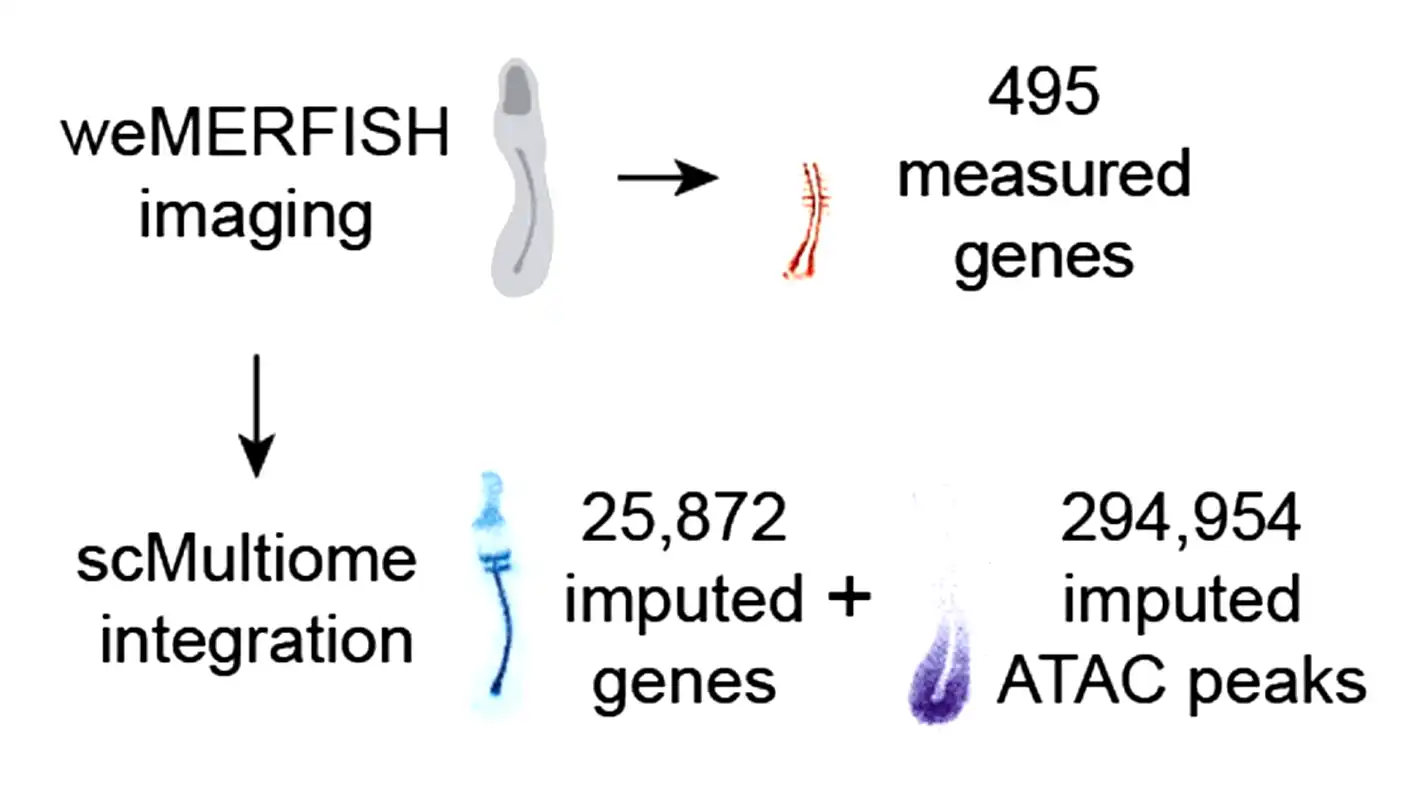
A research team at the University of Basel in Switzerland recently published its findings in the journal Science. They successfully visualized the simultaneous activity of thousands of genes throughout a complete zebrafish embryo. Using this new technology, the team built a massive, comprehensive atlas that maps every gene and cell involved in turning a basic egg into a living organism.
Understanding the interplay between genes and cells during early development is incredibly complex. In the past, scientists could only capture gene activity by slicing the tissue into flat, two-dimensional images. This older method made it impossible to see the whole embryo at once. It also offered very limited spatial detail, meaning researchers often missed the crucial patterns happening inside individual cells.
Professor Alex Schier and his team at the Biozentrum solved this problem. Their new method, called weMERFISH, allows them to measure the activity of nearly 500 genes directly inside intact, three-dimensional tissues. By combining these fresh measurements with older single-cell data, the researchers calculated the spatial patterns of thousands of genes. They also mapped the activity of roughly 300,000 regulatory regions that tell those genes when to turn on and off.
Dr. Yinan Wan, the lead author of the study, explained the core mystery they wanted to solve. “A central question has been: How do thousands of genes work together in an embryo, and how is their activity linked to the movement of cells?” Wan said. The new atlas finally provides the answers, offering a four-dimensional look at early life.
The resulting images are much more than just static photographs. They allow scientists to track processes across both space and time. For example, while watching the zebrafish tail form, the researchers noticed a clear sequence of development along the body. Immature stem cells are gathered right at the tip of the tail. As they looked further forward along the body, they saw cells becoming increasingly mature, eventually turning into solid muscle.
Wan noted that this arrangement essentially lets you see time moving through physical space. The team also discovered a surprising connection between cell movement and gene activity. The changes happening inside the genes are perfectly aligned with how the cells physically move through the growing embryo.
The new atlas also solved a long-standing mystery about how tissues separate from one another. The researchers used the tool to see exactly how clear boundaries form between different body parts, like the muscle and the backbone. They found a specific zone where the activity of many genes changes dramatically from one side of the boundary to the other.
When the team compared early developmental stages to later ones, they noticed something fascinating. Initially, certain genes were active on both sides of the boundary line. Later on, those same genes only remained active on one side. Furthermore, the cells themselves almost never crossed over that line. Schier explained that these sharp boundaries do not form because cells mix together and then physically sort themselves out. Instead, the cells stay put and simply change their internal genetic programming to match their neighbors.
This breakthrough provides a massive foundation for future biological studies. The researchers made all their data freely accessible to the public through a web platform called MERFISHEYES. They want this atlas to serve as a resource for developmental biologists everywhere.
In the future, Schier and his team plan to investigate even more developmental stages to complete the picture of how vertebrates grow. Ultimately, they want to understand the exact combinations of gene activity and cellular behavior required to build specific organs. As Schier put it, “One day we may find out how many ways there are to build a heart or a spinal cord.”
Source: Science (2026).