Key Points:
- A team from Marburg University grew the longest conductive polymer chains ever recorded, reaching nearly 1,000 nanometers in length.
- The new chemical process builds molecules from the ground up without creating messy waste products.
- Researchers used advanced microscopes to observe individual chemical bonds forming during the experiment.
- This breakthrough provides new building blocks for future electronics, like organic transistors and semiconductors.
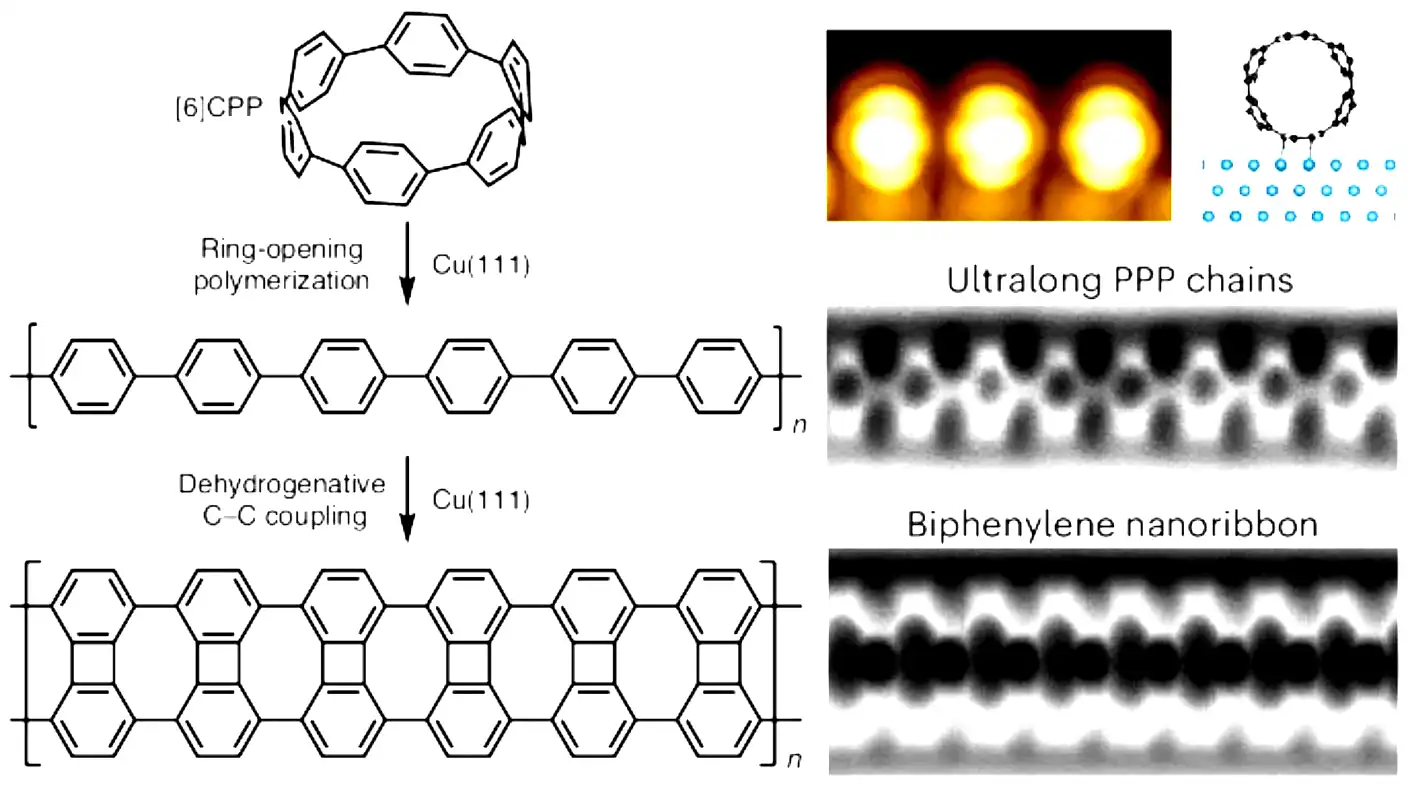
Scientists in Germany just broke a major record in the world of microscopic chemistry. A research team led by Professor Michael Gottfried at Marburg University successfully grew the longest chains of a conductive polymer ever created. These tiny, invisible wires reached lengths of almost 1,000 nanometers, which is roughly one-thousandth of a single millimeter. While that sounds incredibly small to the average person, it is almost 10 times longer than anything scientists could previously build.
The research focuses on a specific material called poly(p-phenylene), or simply PPP. This material is a conjugated polymer, meaning it can readily conduct electrical current from one end to the other. Because of this special ability, tech companies are very interested in using PPP to build the next generation of tiny electronic devices.
However, building these molecular wires has always been a messy and difficult process. In the past, scientists relied on a method in which many short molecular fragments randomly collided with each other and stuck together on a surface. This old method caused many problems because it left behind chemical waste. These unwanted by-products clogged up the surface and stopped the polymer chains from growing any longer.
Professor Gottfried and his team found a much better way. They developed a specific “ring-opening” reaction that acts like a controlled assembly line. Instead of relying on random crashes, the new method uses strained ring molecules. The reactive end of the growing chain carefully opens these rings and attaches them one by one. Gottfried explained that this new mechanism completely prevents those messy by-products from forming, which keeps the surface clean and allows the chain to keep growing indefinitely.
To prove their new method worked, the team needed actually to see the results. They used incredibly powerful tools, including high-resolution scanning tunneling microscopes and non-contact atomic force microscopes. These machines are so advanced that researchers can look at the surface and visually confirm the individual chemical bonds linking the molecules. On average, they measured chains around 170 nanometers long, but they found one massive outlier that stretched nearly 1,000 nanometers across the surface.
This research represents a massive leap forward for basic science. The team essentially expanded the chemical toolkit that engineers use to build atomically precise carbon structures. Gottfried noted that the electronic properties of PPP depend heavily on how long and how perfect the chain is. By creating these ultra-long, flawless chains, the team provided the perfect starting material for building novel carbon nanoribbons. These ribbons could eventually become the tiny organic transistors that power future computers and smartphones.
This breakthrough did not happen in isolation. It required a massive collaborative effort across different scientific fields. The project brought together experts in chemical design, surface physics, and theoretical math from the Universities of Marburg, Giessen, and Leipzig, along with researchers from China. They all worked together under a joint project called the LOEWE focus.
Professor Gert Bange, the Vice-President for Research, praised this teamwork. He said the project perfectly demonstrates the power of close cooperation between different universities. By combining their unique skills, the scientists managed to construct molecules following a precise blueprint and visualize their formation step by step. This collaborative spirit is exactly what leads to scientific breakthroughs and creates new possibilities for the semiconductor technologies of tomorrow.
Source: Nature Chemistry (2026).