Key Points
- Scientists have discovered a new, non-catalytic role for the protein RFC in DNA replication.
- RFC stays attached to the PCNA clamp and slides along the DNA with the copying enzyme.
- This “trio” ensures that the DNA copying process is fast and reliable.
- The discovery upends decades of textbook knowledge in basic biology.
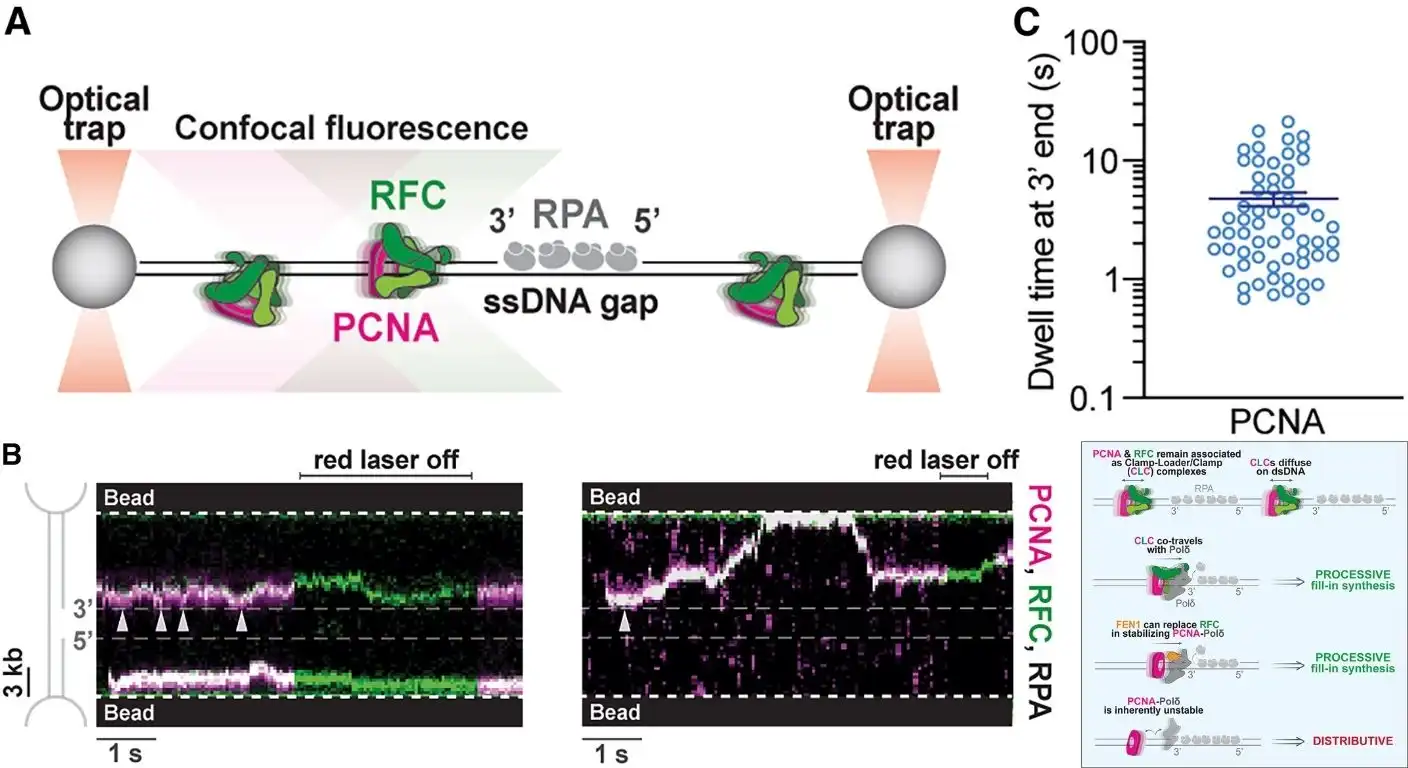
Scientists have made a major discovery about how our cells copy their DNA, a finding that could change our understanding of cancer and neurological disorders. The new research, published in the journal Cell, reveals a surprising new role for a protein called Replication Factor C, or RFC.
For decades, scientists believed that RFC had only one job: to act as a “clamp loader,” snapping a ring-shaped protein called PCNA onto the DNA strand. This PCNA clamp then acts as a tether, holding the DNA-copying enzymes in place so they don’t fall off mid-copy.
But the new study shows that RFC does much more than that. Using a combination of advanced imaging techniques, the researchers found that RFC actually stays attached to the clamp and slides along the DNA as a unit with the copying enzyme. This “trio” ensures that the copying process is both fast and reliable.
“RFC was thought to do its job and leave the scene,” said one of the lead researchers. “But in hindsight, it makes total sense.”
When the team created a mutant version of RFC that couldn’t stay attached, the copying enzyme kept slipping off the DNA, forcing the process to restart over and over again. This dramatically slowed down the replication process and highlighted the crucial, but previously unknown, structural role of RFC.
The discovery is a big deal because the PCNA clamp is a major target for cancer drugs. By understanding how all the different players in the replication process interact, scientists can design more targeted and effective treatments that disrupt tumor growth without harming healthy cells.
The study is also a reminder that even in a well-studied system like DNA replication, there are still new discoveries to be made.
Source: Cell (2026).